(species)

(var. arfakensis)
Papuacedrus papuana
(F. Muell.) H.L. Li 1953
Common names
"Ab (Nega, Kepilam), aiap (Enga), araoem (Karoon), autibo (Kepauko), bit (Yogom), boetsga (Manikiong), bootsjeka (Manikiong), dautie (Kepauko), doewak (Kebar), dzagosa (Asaro, Kefamo), dzasihanini (Asaro, Kefamo), eis (Karoon), gamgan (Hagen, Togoba), gamuga (Hagen, Togoba), hohoba (Lei area), hap (Enga, Kepilam), iwunturra (Manki), juta (Mairi, Watabung), kaibelkombam (Wahgi, Minji), kaipil (Wahgi, Ninj), kamgenkuna (Hagen, Togoba), kap (Enga, Kepilam), matoe (Kepauko), mondalasap (Mendi), mondilasop (Mendi), nipaoe (Kebar), ogeleh (Chimbu, Masul), oleh (Chimbu, Masul), ongol (Wahgi, Minj), pomoan (Manikiong), sowa (Kebar, Andjai), sukou (Wapi, Migote), swa (Andjai), tera (Garaina), toewa (Kebar), urenak (Mendi), wonga (Arfak), ye-enka (Nauti)" (van Royen 1979).
Taxonomic notes
Papuacedrus was initially included in Libocedrus, but was described as a distinct species by Li (1953) primarily on the basis of differences in the female cone, as was usual for generic classification of the Cupressaceae prior to the advent of molecular methods. Molecular studies, however, have tended to refute that analysis, placing Papuacedrus as sister to a clade that contains all the other genera of the Callitroideae (Austrocedrus, Callitris, Diselma, Fitzroya, Libocedrus, Pilgerodendron, and Widdringtonia) (Gadek et al. 2000, Leslie et al. 2012), estimating its divergence from that group at 52 to 72 million years ago, as calibrated by the Eocene species Papuacedrus prechilensis from Argentina (Leslie et al. 2012, also citing Wilf et al. 2009).
Synonymy:
- Libocedrus papuana F. Mueller 1889;
- Thuja papuana Voss 1908;
- Librocedrus torricellensis Schlechter 1913;
- Libocedrus arfakensis Gibbs 1917;
- Papuacedrus torricellensis (Schlechter) Li 1953;
- Papuacedrus arfakensis (Gibbs) Li 1953.
Holotype from Owen Stanley Range (van Royen 1979). This is the sole species in Papuacedrus Li 1953 (syn: Libocedrus Endl. 1847; Austrocedrus Florin et Boutelje 1954) (van Royen 1979).
Van Royen (1979) and all subsequent authorities regard Papuacedrus as a monotypic genus. Li (1953) segregated two additional species (listed in synonymy above). Van Royen argues that the observed variations are due to phenotypic plasticity that is expected in a taxon with such a wide range of habitats. Farjon (2010) recognizes two varieties, the type and P. papuana (F. Muell.) H.L. Li var. arfakensis (Gibbs) R.J. Johns 1995 (syn: Libocedrus arfakensis Gibbs 1917, Papuacedrus arfakensis (Gibbs) H.L. Li 1953, Libocedrus papuana F. Muell. var. arfakensis (Gibbs) de Laub. 1988).
Description
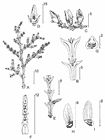
Trees 16-50 m tall and 40-60 cm dbh, rarely shrubs of 1-3 m; normally with a single slender trunk and an open, conical to pyramidal crown of horizontally spreading and ascending branches; crown may be flattened and umbellate at higher altitudes. Outer bark black, blackish grey, greyish brown, or brown, longitudinally grooved, slightly or not peeling, sometimes deeply grooved and strongly peeling with small scales or long fibres. Inner bark pinkish brown, or straw-coloured, becoming white towards centre, with some yellowish resin. Sapwood light brown, yellow or white. Heartwood purple brown, reddish brown, brown, or light brown. Foliage branches mostly in flattened sprays; branching frond-like with ultimate twigs gradually shorter, twigs slender, flattened, glabrous, entirely covered with flattened leaves. Leaves light green, or dark olive above and greener below, sometimes greyish silvery, or glaucous on underside. Leaves on lateral twigs scale-like, in whorls of 4; or else decussate, imbricate, facials smaller than laterals. Facials are ca. 1 mm on mature foliage, but up to 8 mm on whip shoots of young plants, rhombic to lanceolate. Laterals are 2-3 mm on mature foliage, up to 20 mm on whip shoots of young plants, lanceolate to oblong. Stomata in two broad bands on the underside of the leaf. Male twigs terminal, 6-12 mm long, with 8-30 microsporophylls in a cylindric, 4-25×2-3 mm pollen cone, each microsporophyll shield-like, peltate, 1-1.5 mm across, acute, obtuse, or rounded, stalk 0.5-1 mm long. Female twigs (sub)terminal, 2-10 mm long with a few leaves below cone, outer scales of cone oblong to ovate, or ovate-elliptic, 4-7 by 2-5 mm, acute or shortly acuminate, at base connate to an oblong, shield-like or ovate, 1-2.5 by 0.5-2 mm large bract, which is acute or obtuse at tip, tip curving outwards and often hook-tipped on inside. Inner scales of cone lanceolate, broadly elliptic, sometimes elliptic-ovate, 6-12 by 2-5 mm, obtuse or acute, on outside up to the middle connate with a triangular or ovate bract, tip of latter obtuse or acute, curving outwards; all scales at first yellowish green, often glaucous, but in the ripe fruit becoming brown, blackish brown or black, often with ridges radiating from tip of bracts either on both sides or on outside only. Seeds elliptic, often oblique, 1.5-2.5×1-2 mm, with a broadly oblong, often oblique, 4-6×2-3 mm, rounded or obtuse wing with many either radiating or parallel nerves (van Royen 1979, Farjon 2010).
The two varieties can be distinguished only by reference to the juvenile foliage and the pollen cones. In var. papuana the lateral pair of transitional leaves on young plants spreads widely from base, up to 6 mm at the farthest point below apex, leaves falcate, apex turned upwards or slightly recurved; pollen cones are 6-15 mm long with up to 16 microsporophylls. In var. arfakensis, the lateral pair of transitional leaves on young plants spreads slightly from base, up to 3 mm at the farthest point below apex, leaves apically recurved toward base of the following pair; pollen cones are 15-25 mm long with up to 30 microsporophylls (Farjon 2010).
Distribution and Ecology
New Guinea and Maluku (the Moluccas). Regional distribution in New Guinea is as follows:
W New Guinea: "Aifat Valley, Tobi Mts, Kebar Valley, Mt Nettoti, Arfak Mts, Mt Genofa, Wissel Lakes area, Mt Doorman, Ilaga, Otomona Valley, Mt Goliath, Hellwig Mts, Mt Agathodaemonis, Idenburg River area, Lake Habbema area, Mt Wihelmina area, Star Mts, Cycloop Mts.
"TNG. Torricelli Mts, Star Mts, Hindeberg Range, Victor Emanuel Mts, McNicoll-Andabare Plateau, Ambum Valley, Wabag, Mt Hagen area, Kerowagi area, Wahgi-Jimmy Divide, Kubor Range, Nondugl area, Mt Wilhelm area, Mt Otto, Daulo, Finisterre Mts, Sarawaket Mts, Cromwell Mts, Mt Amungwiwa, Watut Valley, Bulolo-Wau area.
Papua: "Kaugel Valley, Mt Ambua, Mt Kerewa, Mt Giluwe, Mt Jalibu, Mt Dickson, Owen Stanley Range, Mt Knutsford, Wharton Range, Mt Albert Edward, Mt Victoria, Milne Bay district, Mt Maneau, Mt Dayman.
"A widespread, variable species from the upper lowland forests to the subalpine shrubberies where it is often noticeable as one of the few trees emergent over the shrubberies, 800-3800 m. Flowers the whole year round with probably a main flowering period between March and September, much depending on the altitude and local position" (van Royen 1979).
The type variety is widespread in New Guinea, as noted above. Var. arfakensis occurs in Papua in the Vogelkop Peninsula and the Sudirman Mountains, and on Bacan and Obi Islands in Maluku (Farjon 2010).
This species has an elevation range of (620-)900-3,600(-3,800) m, and is most abundant in cloud forest (precipitation ca. 4,000 mm/yr) at elevations from ca. 1,500 m to the treeline. This is one of the wider elevational ranges to be found in the conifers, and it comes with a variety of ecological settings. In the lower montane rainforest Papuacedrus is usually an emergent tree in a highly speciose rainforest that becomes less complex with elevation. It can occur in a conifer-dominated forest, e.g. with Dacrydium spp., Podocarpus spp., Phyllocladus hypophyllus, and Araucaria cunninghamii var. papuana. The conifer stands are often surrounded by fire induced grasslands (dominated by Imperata cylindrica), ericaceous shrubs, or tree fern grassland. At lower elevations it is typically on basic soils; higher, soils are often acidic swamps, e.g. Papuacedrus-Phyllocladus-Podocarpus woodland over a Gahnia tussock understory (Farjon 2010, citing Johns 1995).
The type variety is widespread and still abundant, and is not of conservation concern. Variety arfakensis is less abundant and occurs in two disjunct areas, one of which (Wissel Lakes area of Papua) is subject to increasing development pressure; accordingly, its conservation status is "near threatened" (Farjon 2010).
Zone 10 (cold hardiness limit between -1°C and +4.4°C) (Bannister and Neuner 2001).
Remarkable Specimens
No data as of 2023.03.03.
Ethnobotany
"The timber is rather extensively used for building purposes, while the bark is used for roofing" (van Royen 1979). Farjon (2010) reiterates this assessment, noting that the species is rarely planted and is very rare in horticulture, only recorded at a few botanical gardens.
Observations
Remarks
The epithet papuana refers to Papua, synonymous with New Guinea.
Plants of this genus have been found in Eocene sediments of Argentina (Wilf et al. 2009), late Eocene-Oligocene (about 38 million years ago) sediments of Tasmania, and in early Miocene (about 20 million years ago) sediments in southern New Zealand (Pole 2007).
Citations
Johns, R. J. 1995. Papuacedrus papuana var. papuana, Cupressaceae. Curtis's Botanical Magazine 12(2):66-72.
Li Hui-Lin. 1953. A reclassification of Libocedrus and Cupressaceae. Journal of the Arnold Arboretum 34:17-34, pl. I-II. Available: Biodiversity Heritage Library, accessed 2021.12.19.
Wilf, Peter, Stefan A. Little, Ari Iglesias, María del Carmen Zamaloa, María A. Gandolfo, N. Rubén Cúneo, and Kirk R. Johnson. 2009. Papuacedrus (Cupressaceae) in Eocene Patagonia: a new fossil link to Australasian rainforests. American Journal of Botany 96:2031-2047. Available: doi.org/10.3732/ajb.0900085, accessed 2021.12.19.
See also
Farjon (2005) provides a detailed account, with illustrations.