Conservation Status

(subsp. grandicona)

(subsp. mertensiana)
Tsuga mertensiana
(Bong.) Carrière (1867)
Common names
Mountain hemlock, black hemlock, hemlock spruce (Peattie 1950), pruche de Patton (Taylor 1993).
Taxonomic notes
Two subspecies:
- Tsuga mertensiana subsp. grandicona. Type: USA, California, Mammoth Lakes, B. Willard 54 (holotype RM).
- Tsuga mertensiana subsp. mertensiana. Type not designated.
There are many synonyms; see POWO for the full list. None are in common use. I also place into synonymy with subsp. mertensiana the putative hybrid with T. heterophylla, Tsuga × jeffreyi (A.Henry) A.Henry 1919. The entire existence of this hybrid seems to be founded on the initial description by Henry and Flood (1919), based upon two trees grown from seed collected in habitat, and upon a chemotaxonomic study of trees in habitat by Taylor (1972) that concluded "hybridization of Tsuga heterophylla and T. mertensiana is a possible but rare phenomenon and is not necessarily indicated by morphological intermediacy." After nearly a half-century of living in T. mertensiana habitat and talking with botanists and others familiar with the species, I have yet to meet anyone who can point to a hybrid tree in habitat, and hybridisation has never been verified experimentally (Taylor 1972, Farjon 1990, Taylor 1993). The weight of evidence is that these species do not hybridize even though they sometimes grow together (in the upper montane/lower subalpine zone of closed-canopy conifer forest) and molecular studies have shown them to be sister taxa within the clade of North American hemlocks (Feng et al. 2021).
Description
Trees to 200 cm dbh and 55 m tall, typically with a single straight trunk and a dense, narrowly conical crown; but often dwarfed and contorted in snowy subalpine settings, and forming a krummholz in alpine areas. Bark first rough, medium gray, but on stems larger than about 10 cm thick becoming very rough, longitudinally furrowed, dark gray or dark reddish brown. Twigs yellowish or orange-brown, densely pubescent, turning gray in 2-4 years, ridged with decurrent pulvini and oval leaf scars. Foliage buds ovoid-conical, 2-3 × 1-2 mm, brown, not resinous. Leaves densely cover the twig, spreading in all directions, ascending above shoot, 7-20 × 1-1.5 mm, short-petiolate, linear, straight to curved, thick (may have 2, 3 or 4 apparent sides), shallowly grooved at base, apex obtuse to acute, bearing stomata on all surfaces, green and sometimes glaucous. Pollen cones pendulous, ca. 10 mm long, purplish blue ripening yellow. Seed cones mostly borne in upper crown, erect becoming pendant, short-pedunculate or sessile, ovoid-oblong to cylindrical, 3-8.1 × 1.5-3.3 cm when scales are opened, purplish blue ripening light to dark brown, falling a few months after seeds are shed. Fertile seed scales usually 40-72, obovate-cuneate, larger ones 10-18 × 7-15 mm, wide-spreading in mature dry cones, outer margin rounded and entire, with included bracts 4-7 mm long. Seeds cuneate, 3-5 × 2-2.5 mm, brown, with a 4-7 mm light brown wing. 2n=24 (Taylor 1993, Farjon 2010, and pers. obs. of trees in habitat). See García Esteban et al. (2004) for a detailed characterization of the wood anatomy.
In the type subspecies, the seed cones are 3-5.5 cm long with 50-72 fertile scales, and the dark brown scales are 10-13 × 7-10 mm. In subsp. grandicona, the seed cones are 3.5-8.1 cm long and 1.9-3.3 cm wide when opened; there are 40-52 mature seed scales, and the light brown scales are 12-18 × 10-15 mm (Farjon 2010).
Distribution and Ecology
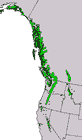
Canada: British Columbia; USA: Alaska, Washington, Idaho, Montana, Oregon, California and Nevada at 0-3050 m. The elevational range of the species varies with latitude, ranging from 0-1070 m in Alaska, to 1600-2300 m in southern Oregon, to 2750-3050 m in the southern Sierra Nevada. Subsp. mertensiana occurs north, and subsp. grandicona south, of the Siskiyou-Klamath mountains near the Oregon-California border. The transition between subspecies is gradational. See also Thompson et al. (1999). Hardy to Zone 4 (cold hardiness limit between -34.3°C and -28.9°C) (Bannister and Neuner 2001).
Distribution data from GBIF Occurrence Download https://doi.org/10.15468/dl.nj5wtr. Red icons are the type subspecies and blue, subsp. grandicona. Green shows the introgression zone in the Klamath and Siskiyou Mountains. Click on any icon for more information.
Throughout the range of this species, mean annual temperature is approximately constant at 3-4°C and precipitation, though highly variable (970 to 3020 mm), does not show a latitudinal gradient. However, the portion of precipitation falling as snow decreases greatly with latitude, from 88 percent in California to 14 percent in Alaska, reflecting a translation from Mediterreanean to subarctic maritime climates. Mountain hemlock's habitat is relatively coastal; it does not occur in the rainshadow of the Coast-Cascade-Sierra Nevada mountain chain except locally in the Rocky Mountains where coastal airmasses can penetrate and produce high winter snowfall. It has been suggested that the species' affinity for areas with persistent winter snowpacks is because it cannot tolerate frozen soils. Throughout most of its range, T. mertensiana defines the upper portion of the subalpine forest and is conventionally divided into two elevational zones: the forest subzone of continuous closed-canopy forest, and the parkland subzone of tree clumps separated by fellfields, wet meadows or low subalpine shrubs. Within the parkland subzone, the discontinuous forest canopy typically reflects reduced seedling establishment success associated with a deep and persistent winter snowpack. At the highest elevations, T. mertensiana grows as a timberline tree in krummholz form (Arno and Gyer 1973, Burns & Honkala 1990, Taylor 1993). In the forest subzone, common associates include Tsuga heterophylla at lower elevations, as well as Abies amabilis, A. lasiocarpa, A. magnifica, Callitropsis nootkatensis, Larix occidentalis, Picea glauca, P. sitchensis, P. engelmannii, Pinus albicaulis, P. contorta, P. monticola, Pseudotsuga menziesii, and Thuja plicata (Burns & Honkala 1990, Farjon 2010). Most of these associates also occur in the parkland subzone.
Two photos at right illustrate some of the principal features of T. mertensiana tree clumps in the subalpine parkland. These clumps often contain one or two exceptionally large and old trees, along with several younger trees. The younger trees commonly represent several different age classes. The oldest tree may not date the origin of the tree clump; often the decayed remains of still older trees can be found within the clump. The origin and development of these clumps has been studied by various authors; a literature review appears in my dissertation (Earle 1993). Arno and Hammerly (1984) also provide an informative discussion of the subject. Typically these clumps are located on topographic high points where winter snows melt out marginally earlier than on adjacent microsites. Establishment of trees on these sites is commonly correlated with episodes of relatively warm, dry climate that last several years; in the Cascades, for instance, such episodes were recorded in the 1930s (Franklin et al. 1971) and again in the latter 1990s; under global warming, reduced snowpacks are prevalent through the species' range and seedlings and saplings are now commonly seen in formerly treeless meadows (pers. obs. 1990 to 2024). Once a clump contains at least one tree tall enough to project above the winter snowpack, blackbody radiation emitted from the tree causes the snow to melt sooner and faster near the tree than in the open meadow. In this way the tree alters its environment to reduce snow accumulation and produce conditions conducive to both the growth of existing trees and the establishment of new trees, both from seed and via asexual means (layering) (Earle 1993). Over time, the tree clump tends to grow radially; such clumps have been called "timber atolls" (Griggs 1938). The formation of such clumps is not confined to T. mertensiana. Many timberline species have been observed to form clumps in areas where a deep and persistent snowpack retards tree establishment, among them, Abies amabilis, Abies lasiocarpa, Picea engelmannii, Pinus albicaulis, and Callitropsis nootkatensis. It is also not unusual to find more than one species of conifer in a clump. T. mertensiana typically initiates clumps within its range, but such clumps often also support Abies amabilis or Callitropsis nootkatensis. On drier sites, such as in the Rocky Mountains, clumps are commonly initiated by Pinus albicaulis, and a "skirt" of young Abies lasiocarpa then grows up around it. After a period of centuries, the Pinus may die due to shading by the now-tall Abies, and the Abies may in its time give way to more shade tolerant Picea engelmannii. Given enough time and a favorable climate, clumps may eventually coalesce to form a continuous forest, but an episode of severe cold/wet weather or a catastrophic fire can reset the clock, returning the site to an open meadow.
Fire is uncommon in most T. mertensiana forests; fires tend to be rare, stand-replacing events (Tesky 1992). The species is also much less subject to pests and pathogens than most conifers. The principal pathogen affecting mountain hemlock is laminated root rot (Phellinus weirii), which is particularly common in Oregon; a number of other fungal pathogens are also known, but are not usually problematic. The dwarf mistletoe Arceuthobium tsugense occurs throughout mountain hemlock's range, but is uncommon north of the Canadian border. Several insect pests are known, but none cause severe or widespread mortality (Burns & Honkala 1990).
Remarkable Specimens
The largest known living specimen is of subsp. mertensiana; it grows in the Enchanted Valley of Olympic National Park, Washington, and was 195 cm dbh and 55.5 m tall when I measured it on 2024.07.18 (photo at right). This tree was discovered by Robert Wood, author of the Olympic Mountains Trail Guide, in 1993. The measurements are somewhat approximate; the height was measured using the "Arboreal Tree" app which has an undescribed methodology but probably uses the tangent method, which is prone to overestimation. The largest tree in Oregon is also subsp. mertensiana; recorded from a 2001 measurement of the Fall Creek tree in Three Sisters Wilderness, it was 207 cm dbh and 34.1 m tall (Black 2019). The largest recorded in California (subsp. grandicona) is between Upper and Lower Cathedral Lakes in Yosemite National Park, and was 197 cm dbh and 27.7 m tall when measured in 2022 (UFEI 2024). The thickest tree ever recorded, also subsp. grandicona, was found in 1951 in Alpine County, California; it was 34 m tall with a dbh of 224 cm and crown spread of 13 m, but died in 2000 (American Forests 1996 and pers. comm. Van Pelt 2024.07.21). The tallest tree on record was 59.1 m tall with a dbh of 127 cm when last measured in 1998, in Olympic National Park, WA (Van Pelt [who measured this tree] e-mail 1998.03.18). I have seen, but not precisely measured, other trees nearly big; for instance, while hiking the Pacific Crest Trail in Washington in 2021, I saw a tree approximately 200 cm dbh and 20 m tall at Wards Pass.
John Muir (1894) described a tree 19 feet 7 inches in girth (191 cm dbh) near Lake Hollow in the Sierra Nevada (subsp. grandicona).
Parish and Antos (2004) analyzed age structures for mountain hemlocks in northern Vancouver Island, British Columbia (subsp. mertensiana) and found one specimen 1059 years old (measured and crossdated); this was from a stump in a clearcut. The same site also produced some of the oldest known Abies amabilis and Callitropsis nootkatensis (also stumps in a clearcut). Another very old living specimen, 722 years, was documented in a tree-ring chronology covering the period 1188-1990 (crossdated after 1370), collected in the Three Sisters Wilderness, Oregon, by Emily Heyerdahl (doi.org/10.25921/hthn-eb42).
Ethnobotany
The wood of Tsuga mertensiana is somewhat inferior to that of T. heterophylla as timber and as pulp, a fact that has little retarded extensive logging of subalpine forests. The species is adaptable to a wide variety of climatic conditions and is widely used as an ornamental (Burns & Honkala 1990) (USDA hardiness zone 5). In this capacity it differs from many alpine conifers in its tolerance of relatively warm, damp environments, and in such settings remains healthy while growing slowly; these are ideal characteristics for an ornamental conifer.
This species has been useful in dendrochronology. As of 2024, there have been about 45 published studies dating as far back as 1923. The great majority of these studies examined climate or some factor closely related to climate, such as timberline fluctuation or glacier expansion; other studies have examined forest age structure and various ecological settings. The utility of the species for climate studies is due to its occurrence at the alpine timberline and its strong interaction with snowpack accumulation. For instance, it is by now generally accepted that widespread invasion of subalpine meadows in the Pacific Northwest happened during the 1930s and 1940s in response to a prolonged episode of reduced winter snowpacks (Franklin 1988). Graumlich and Brubaker (1986) looked at the relationship between climate and ring width for some stands in the Cascade Range of WA, and I did some (unpublished) exploratory studies focusing on population age structures and competitive interactions in subalpine parklands of the Cascade Range of BC, WA and OR.
Observations
Because this species grows to the alpine timberline, its most picturesque qualities are displayed in high mountain areas. Notably good sites are the parklands, where the landscape is covered by a mosaic of meadow and tree clumps. Examples include the high country of Garibaldi Provincial Park in BC, the Seven Lakes Basin of Olympic National Park in WA, and Jefferson Park in the Mount Jefferson Wilderness of OR. Subsp. grandicona can also be found growing nearly everywhere in the high alpine lake country of the Sierra Nevada, including areas in Yosemite, Kings Canyon and Sequoia National Parks.
Remarks
The epithet honors Karl Heinrich Mertens (1796-1832), a German botanist remembered primarily for collections he made as botanist on the Lutke around-the-world expedition, which included this species. Though Mertens died shortly after his return to Europe, his plant collections were cataloged by A. H. von Bongard, who named this species in his honor. For more about Mertens, see Mirov (1954).
Citations
American Forests 1996. The 1996-1997 National Register of Big Trees. Washington, DC: American Forests.
Arno, Stephen F. and Jane Gyer. 1973. Discovering Sierra trees. Yosemite Natural History Association. 89pp.
Arno, Stephen F. and Ramona Hammerly. 1984. Timberline: mountain and arctic forest frontiers. Seattle: The Mountaineers.
Black, Jerry. 2019. Oregon Big Tree & Shrub Measurements. Tualatin, OR: Jerry Black.
Farjon, Aljos. 1988. Taxonomic notes on Pinaceae 1. Proc. Konin. Ned. Akad. Wetensch. ser. C Bot., 91: 31-42.
Farjon, Aljos. 1990. Pinaceae: drawings and descriptions of the genera Abies, Cedrus, Pseudolarix, Keteleeria, Nothotsuga, Tsuga, Cathaya, Pseudotsuga, Larix and Picea. Königstein: Koeltz Scientific Books.
Feng, Yuan-Yuan, Ting-Ting Shen, Cheng-Cheng Shao, Hong Du, Jin-Hua Ran, and Xiao-Quan Wang. 2021. Phylotranscriptomics reveals the complex evolutionary and biogeographic history of the genus Tsuga with an east Asian-North American disjunct distribution. Molecular Phylogenetics and Evolution 157:107066. https://doi.org/10.1016/j.ympev.2020.107066.
Franklin, J. F., W. H. Moir, G. Douglas, and C. Wiberg. 1971. Invasion of subalpine meadows by trees in the Cascade Range. Arctic and Alpine Research 3(3): 215-224.
Franklin, Jerry F. 1988. Pacific Northwest forests. P. 103-130 in M.G. Barbour and W.D. Billings, eds., North American terrestrial vegetation. Cambridge: Cambridge University Press.
Graumlich, Lisa J. and Linda B. Brubaker. 1986. Reconstruction of annual temperature (1590-1979) for Longmire, Washington, derived from tree rings. Quaternary Research 25:223-234.
Griggs, R. F. 1938. Timberlines in the northern Rocky Mountains. Ecology 19:548-564.
Havill, Nathan P., Christopher S. Campbell, Thomas F. Vining, Ben LePage, Randall J. Bayer, and Michael J. Donoghue. 2008. Phylogeny and biogeography of Tsuga (Pinaceae) inferred from nuclear ribosomal ITS and chloroplast DNA sequence data. Systematic Botany 33(3):478–489.
Henry, Augustine, and Margaret G. Flood. 1919. The history of the Dunkeld hybrid larch, Larix eurolepis, with notes on other hybrid conifers. Proc. Roy. Irish Acad. 35(B): 55-66. Available: Biodiversity Heritage Library, accessed 2024.07.21.
Mirov, N. T. 1954. Lodgepole pine discovered and misnamed. Madroño 12:156-157. Available: Biodiversity Heritage Library, accessed 2023.07.17.
Muir, John. 1894. The Mountains of California. Available: http://yosemite.ca.us/john_muir_exhibit/writings/the_mountains_of_california/index.html, accessed 2019.02.04.
Parish, Roberta, and Joseph A. Antos. 2004. Structure and dynamics of an ancient montane forest in coastal British Columbia. Oecologia 141(4):562-576.
Taylor, R. J. 1972. The relationship and origin of Tsuga heterophylla and Tsuga mertensiana based on phytochemical and morphological interpretations. American Journal of Botany 59: 149-157.
Tesky, Julie L. 1992. Tsuga mertensiana. In: Fire Effects Information System. https://www.fs.usda.gov/database/feis/plants/tree/tsumer/all.html, accessed 2024.07.21.
See also
Brooke, R. C., E. B. Peterson, and V. J. Krajina. 1970. The subalpine mountain hemlock zone. Ecology of western North America 2:151-307.
Elwes and Henry 1906-1913 at the Biodiversity Heritage Library (as T. pattoniana) (Photo). This series of volumes, privately printed, provides some of the most engaging descriptions of conifers ever published. Although they only treat species cultivated in the U.K. and Ireland, and the taxonomy is a bit dated, still these accounts are thorough, treating such topics as species description, range, varieties, exceptionally old or tall specimens, remarkable trees, and cultivation. Despite being over a century old, they are generally accurate, and are illustrated with some remarkable photographs and lithographs.
Lanner 1983.
MacKinnon et al. 1992.
Parsons, D. J. 1971. The southern extensions of Tsuga mertensiana (mountain hemlock) in the Sierra Nevada. Madroño 21: 536-539. Available: Biodiversity Heritage Library, accessed 2021.12.19.
Sargent (1898) provides an exceptionally detailed description of this species, with an excellent illustration.