
One of the biggest trees, newly discovered [Phil Barker, 2020.01.27].

Old canopy emergent tree about 40 m tall near Mangamate Hut, Whirinaki Forest [C.J. Earle, 2003.03.05].

Canopy emergent tree on the Ohinetaki Track [C.J. Earle, 2003.03.09].

Young rimu about 6 m tall growing in a field near the start of the Waihoanga Gorge Loop Track, Puketi Forest [C.J. Earle, 2003.03.17].
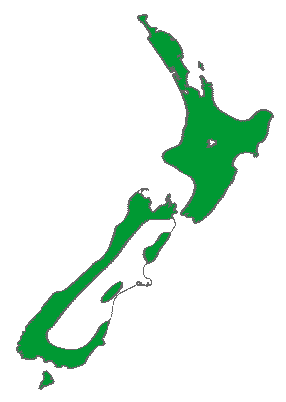
Distribution map (Metcalf 2002).

Looking into the canopy of a big rimu, from the canopy tower at Pureora Forest Park [C.J. Earle, 2003.03.10].

Emergent rimu commonly support a high epiphyte biomass [C.J. Earle, 2003.03.09].

Lower trunk of a tree in Te Urewera National Park [Trevor Hinchliffe].

Foliage on a shade-grown rimu sapling at Kaueranga Visitor Center [C.J. Earle, 2003.03.01].

Sun foliage on the open-grown rimu sapling shown above [C.J. Earle, 2003.03.17].

Bark of a 5 cm diameter rimu at Kaueranga Visitor Center. [C.J. Earle, 2003.03.01].

Bark of an old tree in Whirinaki Forest, showing the distinctive ridging that readily distinguishes rimu from other trees in the native forest [C.J. Earle, 2003.03.06].

Detail photograph
[J.H. Johns].

Detail photograph
[J.H. Johns].

815 rings have been counted on this specimen in the Auckland Museum, sawn from a 250 cm diameter tree felled in the King Country in the 1920s [C.J. Earle, 2003.03.11].

Rimu-dominated forest at Te Urewera National Park [Trevor Hinchliffe].

Rimu logging.

Dacrydium cupressinum
Solander ex G. Forst. 1786
Common names
Rimu, red pine (Allan 1961).
Taxonomic notes
Syn.: Thalamia cupressina (Sol. ex G. Forst.) Spreng. 1826 (Farjon 1998). "In wood anatomy and chemical constituents, D. cupressinum shows such close affinities with species of Podocarpus that it may rightfully belong to that genus" (Dallimore et al. 1967).
Description
Dioecious trees 20-35(-55) m tall and up to 150 (or more) cm dbh. Crown pyramidal when young, with graceful pendulous branches, but acquiring a round-topped crown with strong lateral branches bearing pendulous branchlets and foliage when mature. Bark dark brown or grey, stringy, usually pimpled, and peels in long, thick flakes. The light green leaves on juvenile plants overlap all around the branchlet and are sharply pointed, keeled, linear-subulate, subfalcate, decurrent, 4-7 mm long by 0.5-1 mm wide. This stage persists for many years before beginning to assume the adult form. As it assumes adulthood the leaves become smaller, more closely set and more appressed to the branchlet. They are then about 3-4 mm long, rhomboid. Adult leaves are more appressed, 2-3 mm. long, rigid, subacute, subtrigonous. During the winter the leaves of juveniles may turn bronze in colour but become green again in spring. The adult leaf growing tips are also often bronze-coloured. Pollen cones are small, green, solitary or paired, terminal on branchlets, 0.5-1 cm. long; the apiculus is ovate-acuminate. The ovules are solitary, terminal on curved branchlets, the ultimate leaves forming a swollen, red, succulent (rarely dry) receptacle. Seed is set only at irregular intervals, usually of five to six years. Seeds are about 4 mm. long, oblong-ovoid or narrow-ovoid, little compressed. Wood is dark red (Allan 1961, Dallimore et al. 1967, Salmon 1996, Metcalf 2002).
Always mycorrhizal; forms vesicular-arbuscular mycorrhizas (Hurst et al. 2002).
Distribution and Ecology
New Zealand: N, S and Stewart Is., at 0-600 m elevation. Within its range, mean annual temperature is 10.4°C, with an average minimum in the coldest month of 1.2°C, and a mean annual precipitation of 2633 mm (Biffin et al. 2011, Table S5).
Rimu is usually found in lowland and montane forest, but also occurs in subalpine scrub on Stewart Island (Allan 1961). It is a true rainforest tree and in some forests is the predominant canopy tree, while in others it is more scattered and may be seen only as an emergent tree (Metcalf 2002). "In most forests the distinctive crowns of the mature rimus emerge above the level of the surrounding mixed podocarp-broadleaf forest. Rimu usually occurs in mixed forests in association with other podocarps, hinau or pokaka, tawa and beech. A forest type usually referred to as rimu pole forest occurs on flat, wet terraces on the West Coast of the South Island and on Stewart Island. There are extensive rimu stands from North Okarito Forest southwards ... Elsewhere, it will form a fine specimen tree when growing in the open space of a forest clearing, sheltered field or lawn" (Salmon 1996).
Zone 9 (cold hardiness limit between -6.6°C and -1.1°C) (Bannister and Neuner 2001).
June (1983) studied regeneration in a rimu forest and concluded that regeneration takes place continually beneath an intact forest canopy. Initially very large numbers of seedling are produced, which experience very high mortality within a few years. The surviving sapling and small trees may persist beneath the canopy for 100 to 400 years, during which time they grow exceedingly slowly. When a gap-phase event such as windthrow or death of an overstory tree creates a localized opening in the canopy, the pole-size trees may be able to exploit the improved light environment and finally become canopy codominant trees (June 1983). This is very similar to the life history displayed by trees in another temperate conifer rainforest, that of northwestern North America, where this life history strategy has been described for species such as Tsuga heterophylla and Abies amabilis, both in the Pinaceae.
Beveridge (1964) describes the dispersal and fate of seeds from New Zealand (Pureora Forest) podocarps. He reported two mast years for rimu during a 10-year period, with one more year of reduced but significant seed production. A large mature rimu yielded 4 pounds (1.9 kg) of sound seed, representing 200,000 seeds. These ripened from mid-March to May. Most such seeds germinate in the following late spring and summer. Like nearly all podocarps, the rimu is dispersed by birds that eat the seeds, digest the fleshy receptacle, and excrete the seed. Beveridge found that most podocarp seeds were dispersed by New Zealand pigeons
(Hemiphaga novaeseelandiae), tuis (Prosthemadera novaeseelandiae) and bellbirds (Anthornis melanura). Some smaller birds eat the receptacle piecemeal and drop the seed, and thus do not act as agents of dispersal. Although podocarp seeds are very commonly excreted by birds perching in trees, sampling in an area of burned-over forest found that the majority of tree seedlings were podocarps (mainly rimu), clearly showing that birds effectively disperse the seeds into open areas. Seedlings were especially clumped around stumps, which may have been the perches. Beveridge also detailed the destruction of podocarp seed. Among rimu, 80% of seed is apparently destroyed in the tree's crown by animal (insect, bird or rodent) activity. Responsible birds include a variety of non-native species (starling, English sparrow and California quail, and chaffinch, Fringilla coelebs) but only one native species (yellow-crowned parakeet, Cyanoramphus auriceps). Black rats (Rattus rattus), an abundant invasive species, show a hearty appetite for podocarp seeds, preferring rimu above all other species. Among insects, the weta (Hemideina thoracica) has been shown to eat rimu seeds, consuming the seed coat and opening the seed within. These results clearly suggest that non-native species are taking a heavy toll during rimu mast years, but the possible ecological consequences of this are not discussed by Beveridge (1964).
Remarkable Specimens
Currently the largest known tree is one found by Kevin Barker and Olga Brochner on 2020.09.26 in Otanewainuku forest, 258 cm dbh and 42 m tall, and easy to access (photos and further details are at New Zealand Notable Trees, accessed 2020.12.09). This is the largest rimu ever recorded. Unfortunately it appears to be in decline and may not have long to live. The big tree shown at right was found on 2020.01.27 by Phil Barker and Lindsay Molloy, near the central west coast on the South Island. When found, it measured 252 cm dbh and 35.1 m tall and was in excellent health. Photos and further details are at New Zealand Notable Trees (accessed 2020.02.24). A comparable specimen was found on the same day, by the same party, in the same area as the record tree; it is 242 cm dbh and 40.6 m tall (photos and further details are at New Zealand Notable Trees, accessed 2020.12.09), and is described as being in good health. Other huge specimens exist today in some remnants of the once extensive West Taupo forests and in a few North Island reserves such as the margins of the Urewera forests near Minginui and at Opepe Bush near Taupo (Salmon 1996). There is also a historic record of a tree 42 m tall and 253 cm dbh, found near Pureora (Burstall and Sale 1984), but a 2002 effort to locate it was unsuccessful and it is presumed dead.
The oldest tree known in shown in the accompanying photo of a specimen with 815 rings. I know of none older, though as with most large trees, great ages are sometimes alleged with no supporting evidence. Bathgate (1987) refers intriguingly to several hundred stem discs covering the past 700-800 years, and to the existence of multiple chronologies, but I have found no evidence that these data have been published or even utilized. June (1983) reports sampling trees up to 1200 years old via ring counts on basal stem sections, but cites no data. A maximum of 1200 years is also cited by Wardle (1991).
Ethnobotany
See Conifers of New Zealand for a review of the historic role of forests in native and European cultures of New Zealand.
In March 1773 (on his second voyage), Captain James Cook stopped in Dusky Sound to brew a beer he called a spruce beer, intended to combat scurvy, made from sugar (treacle or molasses) fermented with young rimu shoots (Salmon 1996). Such beers, made with young spruce (Picea) shoots, were commonly used for the purpose in England and the New World, due to the high vitamin C content of the young shoots.
Later, it became an important timber tree. "The heavy wood of rimu is deep red in colour, strong, hard and durable - though not so durable as kauri or totara. The heartwood is nearly always beautifully figured, as a result of the grain being twisted during growth. Heart rimu is one of the most beautifully figured woods in the world, and for this reason has always been highly prized in New Zealand as a finishing timber for doors, door frames, panelling and furniture. The timber has also been extensively used in the past for house framing and weatherboarding" (Salmon 1996). Other reported uses include bridge construction, railway sleepers, and posts; and "the bark contains a good deal of tannin and is said to impart a reddish colour to leather" (Dallimore et al. 1967). "With growing scarcity and diminishing cut its use is becoming more restricted. In future the use of rimu will undoubtedly be confined to plywood veneers and other decorative purposes" (Salmon 1996).
The species is scientifically useful through dendrochronology. The earliest work is recounted by Bell and Bell (1958), who dismiss the species as unusable. The species poses problems because is subject to ring wedging. "Ring wedging occurs when rapid radial growth occurs over several years in certain segments of the tree's circumference, while being extremely slow or absent in others... Examination of complete cross sections can help overcome this problem, and for some species accurate age counts must be based on cross sections rather than increment cores" (Norton and Ogden 1990). However, it has been used in at least one climate change study (Burrows & Greenland 1979).
Observations
Of all the New Zealand conifers, this is probably the one you are most likely to find growing as an ornamental tree outside of New Zealand. Within the country, it is extremely widespread, growing as an ornamental, as a recruit in pasture land, and of course as a dominant forest tree in many native forests. Some of the best remaining forest trees can be found in Pureora and Whirinaki Forest Parks. A walk along the Hunter's Track in Pureora, or along the Whirinaki River south of Minginui, will reveal trees more than 200 cm dbh and approximately 40-50 m tall, among the tallest native trees in New Zealand.
Remarks
The epithet indicates a similarity to an unspecified species of cypress. For more detail, see also the Remarks for Dacrydium.
First collected near Queen Charlotte Sound in early 1770 by Banks and Solander, the botanists on Captain James Cook's celebrated first voyage (Salmon 1996).
The cones are rich in bioavailable calcium and vitamin D. The kakapo (Strigops habroptilus), a highly endangered parrot endemic to New Zealand (and the heaviest parrot in the world), has been found to breed successfully only during D. cupressinum mast years. The vitamin D is in the D3 form, formerly thought to be produced only by animals, more recently discovered to be produced by some plants in the Solanaceae; this is the first report of it being produced by a conifer (von Hurst et al. 2016).
"As we have seen, rimu is one of our most ancient trees; fossil pollen grains of trees very similar to D. cupressinium have been traced back 70 million years. Rimu was very common in the podocarp forests that once covered New Zealand. In these forests it was often the dominant tree, sometimes occurring in almost pure stands over small areas" (Salmon 1996).
Ruscoe et al. (2004) document a house mouse (which is invasive in New Zealand) irruption in response to a heavy rimu seedfall event. The seedfall event is itself interesting; the authors allege that "rimu masts two years after a cool summer (the time of floral initiation) and in the year of a warm summer" (Schauber et al. 2002, cited by Ruscoe et al. 2004). It is very unusual to hear mast years predicted with such confidence for any tree species. Ruscoe et al. (2004) go on to show that the mice did in fact exploit the seedfall, and that the nutritional content of rimu seed is more than adequate to support the energy demands of a mouse irruption.
Citations
Allan, H. H. 1961. Flora of New Zealand. Volume I, Indigenous Tracheophyta. Wellington: R.E. Owen Government Printer.
Bathgate, J. 1987. Downturn in rimu (Dacrydium cupressinum) recruitment and diameter growth from 1700-1900 AD. New Zealand Journal of Ecology 10:162.
Bell, V., and R. E. Bell. 1958. Dendrochronological studies in New Zealand. Tree-Ring Bulletin 22:7-11.
Beveridge, A. E. 1964. Dispersal and destruction of seed in central North Island podocarp forests. Proceedings of the New Zealand Ecological Society 11:48-56.
Forster, G. 1786. De Plantis Esculentis Insularum Oceani Australis Commentatio Botanica, p. 80. Available: Biodiversity Heritage Library, accessed 2021.12.24.
June, S. R. 1983. Rimu regeneration in a north Westland hardwood podocarp forest. New Zealand Journal of Ecology 6:144-145.
Hurst, S. E., M. H. Turnbull and D. A. Norton. 2002. The effect of plant light environment on mycorrhizal colonisation in field-grown seedlings of podocarp-angiosperm forest tree species. New Zealand Journal of Botany 40:65-72.
Ruscoe, Wendy A., Deborah Wilson, Lisa McElrea, Gary McElrea, and Sarah J. Richardson. 2004. A house mouse (Mus musculus) population eruption in response to rimu (Dacrydium cupressinum) seedfall in southern New Zealand. New Zealand Journal of Ecology 28(2):259-265.
Schauber, E. M., D. Kelly, P. Turchin, C. Simon, W. G. Lee, R. B. Allen, I. J. Payton, P. R. Wilson, P. E. Cowan, and R. E. Brockie. 2002. Masting by eighteen New Zealand plant species: the role of temperature as a synchronizing cue. Ecology 83:1214-1225.
von Hurst, P. R., R. J. Moorhouse and D. Raubenheimer. 2016. Preferred natural food of breeding Kakapo is a high value source of calcium and vitamin D. Journal of Steroid Biochemistry and Molecular Biology 164:177-179. doi: 10.1016/j.jsbmb.2015.10.017.
Wardle, P. 1991. Vegetation of New Zealand. Cambridge: Cambridge University Press.
See also
Kirk, T. 1877. A revised Arrangement of the New Zealand Species of Dacrydium, with Descriptions of new Species. Transactions and Proceedings of the Royal Society of New Zealand 10:383-391 (accessed 2011.03.27).
Norton, D. A., C. H. Cochrane, and S. D. Reay. 2005. Crown-stem dimension relationships in two New Zealand native forests. New Zealand Journal of Botany 43:673-678.
Smale, M. C., B. R. Burns, P. N. Smale and P. T. Whaley. 1997. Dynamics of upland podocarp/broadleaved forest on Mamaku Plateau, central North Island, New Zealand. Journal of the Royal Society of New Zealand 27: 513-532. Abstract: Forest composition was examined on one hectare of upland Dacrydium cupressinum-Prumnopitys taxifolia/Weinmannia racemosa-Beilschmiedia tawa forest on south Mamaku Plateau, central North Island, New Zealand, a site of catastrophic volcanic disturbance c. 1900 years ago. ... Tree ferns, mostly Dicksonia squarrosa and Cyathea smithii, ... dominated basal area in younger (< 80 yr) forest; D. cupressinum dominated basal area in older (>80 yr) forest. ... Emergent D. cupressinum and P. taxifolia sampled were aged between apprx400 and apprx1000 yr; established seedlings of both species were rare. Much younger Pectinopitys ferruginea occurred commonly but only up to small tree size; Podocarpus totara was present only as long-dead fallen trees. Broadleaved species were all much younger (< apprx 250 yr). Shade-tolerant P. ferruginea, ... regenerate continuously in the understorey of high forest, although P. ferruginea requires some canopy opening to develop beyond sapling size. ... Larger-scale studies over longer time periods and over larger areas are needed to determine definitively the regeneration strategies of the conifers. The unexpectedly high proportion of building-phase forest suggests a period of substantial canopy collapse within the past century. Widespread fallen podocarps on the forest floor indicate the presence of dense conifer forest on the site in the discernible past, ...
Stewart, Glenn H. 2002. Structure and canopy tree species regeneration requirements in indigenous forests, Westland, New Zealand. DOC Science Internal Series 66:5-33. Available: www.doc.govt.nz/upload/documents/science-and-technical/dsis66.pdf, accessed 2009.06.09. This paper discusses, for a variety of New Zealand species, the regeneration requirements of main canopy tree species; the natural gaps that occur in old-growth forests; and the areas affected by natural disturbances as reflected by patch sizes of relatively even-aged groups of trees.
Gymnosperms of New Zealand.
The New Zealand Plant Conservation Network, accessed 2010.11.22.

















