Phyllocladus
L.C. et A. Rich. 1826
Common names
Celery top pine.
Taxonomic notes
A genus of five species:
Syn.: Podocarpus Labill. Nov. Holl. Pl. Sp. 2 (1806) 71, t. 221, non L'Herit. ex Pers. (1807).; Brownetera L.C. Richard, Ann. Mus. Hist. Nat. Paris 16 (1810) 299, nom. nud; Thalamia Spr. Anl. Kennt. Gewächse ed. 2, 2 (1817) 218 (de Laubenfels 1988).
Type species Phyllocladus rhomboidalis L.C. Richard, nom. illeg.=Phyllocladus aspleniifolius (Labill.) Hooker; basionym Podocarpus aspleniifolia Labill., l.c." (Van Royen 1979).
Keng (1962-1978) suggests that this genus should be placed in a family of its own, Phyllocladaceae Bessey 1907. The genus is differentiated from other podocarps in having a structure resembling an aril (see Taxaceae) as well as an epimatium, and in having a different number of chromosomes and a different pollination mechanism. However, it also has much in common with other podocarps; "they share as winged pollen with a prothallial tissue, an epimatium, and binucleate embryo cells. Other significant common traits are a solitary ovule per fertile bract, two pollen sacs per microsporophyll, and fused pairs of cotyledons as well as fleshiness of the mature cone and a mature seed of essentially identical form as those of other genera with naked seeds in the family" (de Laubenfels 1988, citing Singh [Embryology of Gymnosperms, 1978, 257]). The question of whether to assign family status hinges mainly on the question of whether the unusual foliage morphology (phylloclades, discussed below) represents a primitive character linking Phyllocladus to extinct pro-Gymnosperms, or a derived character in an otherwise Podocarp-like genus (Farjon 1998). De Laubenfels (1988) dismisses Keng's hypothesis, noting that "perfectly typical coniferous leaves are produced in the juvenile phase."
Molecular studies, reviewed in the "Taxonomic notes" section of Podocarpaceae, have so far placed Phyllocladus with Lepidothamnus in a basal clade sister to all the other genera in the Podocarpaceae. Work by Wagstaff (2004) using the chloroplast genes rbcL and matK in all 5 Phyllocladus species found a monophyletic clade, nested within other Podocarpaceae genera, and again sister to Lepidothamnus. The genus evidently diverged from the rest of Podocarpaceae fairly early in the family's history (ca. 190 million years ago), but the extant species are all fairly closely related, evidently having originated in a rapid speciation event approximately 6.3 million years ago (Wagstaff 2004).
Description
Trees up to 30 m tall, monoecious but unisexual individuals are common. Bark smooth, dark brown or black, platy, red or yellow and fibrous within, shed in large thin flakes. Crown abundantly branched, primary branches in false whorls and secondary branching is abundant. Foliage: The ultimate foliar shoots are flattened into cladodes or 'phylloclades' which involve a central axis and several alternate side 'shoots'. In outline these cladodes can be oval, triangular, deeply lobed, or compound and small marginal hooks representing reduced leaves can sometimes be seen. Phylloclads reach several cm in length or can be aggregated along branches in complexes to more than 20 cm long or transitional as a large deeply lobed phylloclad. Shoots which are to continue growth, whether a secondary axis or a lobed cladode, terminate in a globular bud formed of overlapping triangular scales. These in turn develop into short shoots covered with linear lanceolate caducous scale-leaves in the axils of which new cladodes or fertile structures may be produced. Seedlings bear spirally arranged, single-veined, linear, acute bifacially flattened leaves up to 1 cm long followed gradually by smaller, more lanceolate forms until the adult scales are produced. Pollen cones cylindrical, clustered each in the axil of a scale of a secondary shoot and each subtended by a short to long, mostly naked stalk and by a few sterile scales. Seed cones appear singly or grouped either terminally or laterally in the axil of a scale on a naked stalk, at the base of a cladode, or terminally or laterally on a reduced or unreduced cladode. The cone consists of a few to many thickened spirally arranged scales, some of which bear a single erect ovule on the upper surface. The developing seed is surrounded to at least half its length by a symmetrical or nearly symmetrical filmy white aril or rough-edged epimatium. Seeds, as many as 20 per cone but usually only 2 or 3, are oval and wider than thick, protrude from the bright red ripe cone, have a crooked micropyle at the tip, and are dark brown to black (de Laubenfels 1969, 1988).
Distribution and Ecology
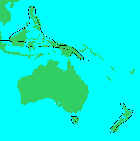
New Zealand; Australia: Tasmania; Papua New Guinea; E Malaysia: Borneo; E India; Philippines (Allan 1961, Silba 1986). It typically occurs in upland tropical and temperate rain-forest, often mossy forest, as a large canopy tree to stunted forms near the tree line (de Laubenfels 1988).
Remarkable Specimens
No particularly large or tall specimens are recorded, but thanks to investigations using dendrochronology, age estimates are available for the Australian and New Zealand species; thus far, the oldest appears to be Phyllocladus aspleniifolius.
Ethnobotany
Several (probably all) species have been exploited for their timber. The four Australian and New Zealand species have all received some attention through dendrochronological research.
Observations
See the species accounts.
Remarks
"Fossils have been reported from the Eocene to Quaternary in New Zealand and from the Oligocene in New South Wales and Victoria (Australia); fossil pollen of Oligocene age was found in Australia, New Zealand and western Antarctica (Couper, Proc. R. Soc. Lond. ser. B, 152, 1960, 491). The Malesian extension of the range was probably only reached in the late Tertiary. It is now extinct in Australia outside Tasmania (Florin, Kongl. Svensk. Vet. Ak. Handl. III, 19, n. 2, 1940, 75, map 4; Acta Horti Berg. 20 (4), 1963, 184, t. 17: map)" (de Laubenfels 1988).
Citations
Allan, H. H. 1961. Flora of New Zealand. Volume I, Indigenous Tracheophyta. Wellington: R.E. Owen Government Printer.
Wagstaff, Steven J. 2004. Evolution and biogeography of the austral genus Phyllocladus (Podocarpaceae). Journal of Biogeography 31:1569-1577.
See also
Keng, H. 1962. Ann. Bot. n.s. 26: 69, 14 fig.
Keng, H. 1963. Gard. Bull. Sing. 20:123, 127.
Keng, H. 1973. Taiwania 18:142.
Keng, H. 1974. Ann. Bot. 38:757.
Keng, H. 1975. Taxon 24:289.
Keng, H. 1977. Pl. Syst. Ecol., Suppl. 1:235.
Keng, H. 1978. The genus Phyllocladus (Phyllocladaceae). Journal of the Arnold Arboretum 59: 249-273.
Molloy, B.P.J. 1999. A contribution to the taxonomy of Phyllocladus (Phyllocladaceae) from the distribution of key flavonoids. New Zealand Journal of Botany 37:375-382. Focuses on the New Zealand species, but primarily of interest for its general discussion of Phyllocladus taxonomy.
Quinn, C.J. 1987. The Phyllocladaceae Keng - a critique. Taxon 36(3):559-565.
Rich., L.C. Ex Mirbel. 1825. Mém. Mus. Hist. Nat. Paris 13: 76 (nom. cons.).
Rich. L.C. and A. Rich. 1826. Comm. Bot. Conif. & Cycad. 129, t. 3, f. 12.
de Laubenfels (1969).
Van Royen (1979).
Gymnosperms of New Zealand.