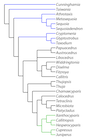
Phylogenetic tree for the Cupressaceae, primarily based on molecular evidence. Branches shown in blue represent genera formerly assigned to the Taxodiaceae; those in green represent the clade of Cupressus sensu latu. Analysis is detailed in Yang et al. (2022), on which this figure is based.

Foliage and old cone of Calocedrus decurrens. The morphology of the female cone is traditionally the basis for generic divisions within the Cupressaceae [C.J. Earle, 2019.06.09].

This Sequoia sempervirens, the Crannell Giant, was the single largest tree ever recorded in all of human history (scanned from Hammond Lumber Company advertisement in American Lumberman, 1911).

Athrotaxis cupressoides about 5 m tall, in alpine heath on the Shelf Tarns track, Mt. Field National Park [C. J. Earle, 2015.02.27].

Callitris pancheri growing near a river [Adam Black, 2020.02.01, Facebook post].

"A magnificent stand of mixed Port Orford white cedar and fir timber located in Curry County, Oregon, on the property of the C.A. Smith Timber Company. A fair example of the mixture of fir and cedar growth on Curry County lands" (American Lumberman 1911).

Cryptomeria Avenue [Sugi Namiki] by Hiroshi Yoshida (1937, woodblock print, 37 cm height). This avenue is in Nikko; it leads from the Sacred Bridge outside Nikko to the Nikko Toshogu Shrine Complex - HERE is a contemporary photograph.

A mature seed cone of Cunninghamia lanceolata [C.J. Earle, 2018.01.05].

Cupressus dupreziana in habitat, in the Sahara desert iNaturalist observation 7367127 [András Zboray, 2011.11.23]

Juniperus osteosperma at Toroweap Overlook in the Grand Canyon [C. J. Earle, 2016.05.03].

Foliage of Libocedrus austrocaledonica [C.J. Earle, 2015.02.25].

Thuja plicata is just one of the many species in Cupressaceae where you don't even stop to measure a tree less than 3 meters in diameter [Adrian Dorst, 1999.12].

Filling the fire scar at the base of the Agassiz Tree in Calaveras State Park is a virtual who's who of Sequoiadendron researchers, including Bob Van Pelt, Steve Sillett, Mike Taylor, Dwight Willard, and Wendell Flint. See the Bookstore for works by these authors [C.J. Earle, 1998.07.14].

Cupressaceae
Gray (1822)
Common names
Cypress family; 柏科 bai ke [Chinese]; Zypressengewächse [German]; Κυπαρισσοειδή [Greek]; ヒノキ科 hinoki ka [Japanese]; 측백나무과 [Korean]; Кипарисовые [Russian].
Taxonomic notes
This is the largest conifer family in terms of genera, and the third-largest in terms of species. In this treatment, there are 28 genera (monotypics shown in bold):
There have been some 21st century changes in the Cupressaceae that may not be widely known, so here's a quick summary; see the linked taxa for details:
- Actinostrobus has been merged into Callitris. See Callitris for discussion.
- Fokiena has been merged into Chamaecyparis. See Chamaecyparis for discussion.
- Neocallitropsis has been merged into Callitris. See Callitris for discussion.
- Pilgerodendron has been merged into Libocedrus. See Libocedrus uvifera for discussion.
- Three genera have been segregated from Cupressus: Callitropsis, Hesperocyparis, and Xanthocyparis. See Cupressus for discussion.
Syn.: Cupressoideae Rich. ex Sweet; Cupresseae Rich. ex Dumort.; Cunninghamieae Zucc. ex Endl. (James L. Reveal email 1998.12.31).
An infrafamilial classification also exists (Yang et al. 2022):
| Subfamily |
Tribe |
Genera |
| Cunninghamioideae Silba |
Cunninghamia |
| Taiwanioideae L.Chu Li |
Taiwania |
| Athrotaxidoideae L.Chu Li |
Athrotaxis |
| Sequoioideae Quinn |
Metasequoia, Sequoia, Sequoiadendron |
| Taxodioideae Endl. ex K.Koch |
Cryptomeria, Glyptostrobus, Taxodium |
| Actinostroboideae Koehne |
Papuacedreae Y.Yang |
Papuacedrus |
| Austrocedreae Y.Yang |
Austrocedrus |
| Libocedreae H.L.Li |
Libocedrus |
| Diselmeae Henkel & W.Hochst. |
Diselma, Fitzroya, Widdringtonia |
| Actinostrobeae Henkel & W.Hochst. |
Callitris |
| Cupressoideae Sweet |
Thujopsideae Henkel & W.Hochst. |
Thuja, Thujopsis |
| Chamaecyparideae Y.Yang |
Chamaecyparis |
| Tetraclineae H.L.Li |
Calocedrus, Microbiota, Platycladus, Tetraclinis |
| Cupresseae Dumort. |
Callitropsis, Cupressus, Hesperocyparis, Juniperus, Xanthocyparis |
The Cupressaceae are found in the fossil record since the Jurassic; specifically, since 197-190 million years ago, represented by the extinct species Austrohamia minuta (Escapa et al. 2008 in Rothwell et al. 2012). The family arose via a whole-genome-duplication (polyploidy) event that separated it, in a clade that included the progenitor of both the Cupressaceae and the Taxaceae, from the remaining conifer lineages (a similar event gave rise to the Pinaceae clade, and to the Welwitschiaceae) (Li et al. 2015).
The family was formerly divided between Cupressaceae sensu strictu (genera with leaves opposite in four ranks or whorled) and Taxodiaceae (leaves mostly alternate). Foliage characters, however, are seldom used as grounds for discriminating between families. All genera treated here have seed cones in which the bract-scale complexes are fused for most of their common length, the 1-20 ovules are erect (but may invert with maturity), and the paired seed wings, if present, are derived from the seed coat (Eckenwalder 1976, Watson and Eckenwalder 1993). Study of plastid (rbcL) DNA sequences has further confirmed the close relationship between the Cupressaceae s.str. and the genera formerly assigned to the Taxodiaceae. As shown in the phylogenetic tree at right, the Cupressaceae sensu strictu form a monophyletic clade, with the various Taxodiaceae genera forming basal branches.
Description
Trees or shrubs, generally resinous and aromatic, monoecious (usually dioecious in Juniperus). Bark fibrous and furrowed (smooth or exfoliating in plates in some Hesperocyparis and Juniperus species). Lateral branches well developed, similar to leading shoots, twigs terete, angled, or flattened dorsiventrally (with structurally distinct lower and upper surfaces in Thuja, Calocedrus, Thujopsis, Fokienia, Libocedrus, Papuacedrus, and to a lesser extent in some other genera), densely clothed by scalelike leaves or by decurrent leaf bases; longest internodes to 1 cm; buds undifferentiated and inconspicuous (except in Sequoia, Metasequoia, Cunninghamia and Juniperus sects. Juniperus and Caryocedrus). Roots fibrous to woody (bearing aboveground "knees" in Taxodium). Leaves simple, usually persisting 3-5(-12) years and shed with lateral shoots (cladoptosic) (shed annually in Taxodium, Glyptostrobus and Metasequoia; leaves with an abscision zone and shed individually in Juniperus Sects. Juniperus and Caryocedrus), alternate and spirally arranged but sometimes twisted so as to appear 2-ranked, or opposite in 4 ranks, or whorled, deltate-scalelike to linear, decurrent, sessile or petiolate; adult leaves appressed or spreading, often differing between lateral and leading shoots (twigs heterophyllous), sometimes strongly dimorphic on each twig (in Thuja, Thujopsis, Libocedrus, Papuacedrus and Calocedrus, and weakly so in some other genera) with lateral scale-leaf pairs conspicuously keeled; juvenile leaves linear, flattened, spreading; often with solitary abaxial resin gland; resin canal present. Pollen cones maturing and shed annually, solitary, terminal (rarely in clusters of 2-5, or to 20 or more in Cunninghamia; axillary in Cryptomeria and Juniperus Sects. Juniperus and Caryocedrus; in terminal panicles in Taxodium and Metasequoia), simple, spheric to oblong; sporophylls overlapping, bearing 2-10 abaxial microsporangia (pollen sacs); pollen spheric, not winged. Seed cones maturing in 1-2 seasons, shed with short shoots or persisting indefinitely on long-lived axes (shattering at maturity in Taxodium), compound, solitary, terminal (rarely in clusters of 2-5, or up to 100 or more in Widdringtonia; axillary in Juniperus Sects. Juniperus and Caryocedrus); scales overlapping or abutting, fused to subtending bracts with only bract apex sometimes free; each scale-bract complex peltate, oblong or cuneate, at maturity woody or fleshy, with 1-20 erect (inverted with age in Athrotaxis, Cunninghamia, Glyptostrobus, Taiwania, Metasequoia, Sequoia and Sequoiadendron), adaxial ovules. Seeds 1-20 per scale, not winged or with 2-3 symmetric or asymmetric wings; aril lacking; cotyledons 2-5 (to 9 in Taxodium) (M.P. Frankis [pers. comm. 1999.02.16], Watson and Eckenwalder 1993).
All Cupressaceae appear to share a vesicular-arbuscular mycorrhiza (Newman and Reddell 1987, Brundrett 2008 and citations therein).
Distribution and Ecology
This is the most widely distributed of all gymnosperm families, occurring in diverse habitats on all continents except Antarctica, but all genera other than Juniperus show strongly relictual distributions, with a large number of localised, rare and endangered taxa. Most of the generic diversity is in the southern hemisphere, but the largest genus, Juniperus, is chiefly north-temperate (Silba 1986, Van Royen 1979).
Worldwide distribution of Cupressaceae, generalized to 1x1 degree cells; data from the BRAHMS database at Conifers of the World, accessed 2021.11.11.
The Cupressaceae are, in general, very much of conservation concern. The IUCN (2020) has identified 6 taxa as critically endangered, 24 as endangered, and 17 as vulnerable, thus 34% of all species in the family are at risk. Principal factors of decline include habitat conversion through agriculture or development, timber harvest, grazing, and climate change.
Remarkable Specimens
Sequoiadendron giganteum, also called "Big tree" is the largest of all trees. Sequoia sempervirens is the tallest of all trees.
Four of the five oldest known non-clonal species on Earth are in the Cupressaceae; only Pinus longaeva is known to grow older (exceeding 5,000 years). The oldest specimens yet found, all of which represent verified, crossdated ages supported by extant wood samples, are:
Ethnobotany
The heartwood of many species of Cupressaceae is resistant to termite damage and fungal decay, and therefore it is widely used in contact with soil [e.g., for fenceposts]. Frank Lloyd Wright preferred Taxodium as a siding for wooden residences, while Sequoia is preferred for lawn furniture throughout the vast suburbs of California. The premier coffin wood of China, Cunninghamia lanceolata, is another member of the family, and Chamaecyparis wood is in similar demand in Japan. Many genera are incorrectly called cedars because their heartwood is as aromatic as that of the true cedars, Cedrus (Pinaceae). Wooden pencils are made from incense-cedar (Calocedrus decurrens) and eastern redcedar (Juniperus virginiana), which is also used for lining "cedar" chests. Wood from species of redcedar (Thuja) is used for roofing shingles and for house siding.
Many Cupressaceae are treated with very high regard by traditional societies. In Japan, Cryptomeria japonica is the national tree, and of the 'five sacred trees of Kiso', four are in this family (Chamaecyparis obtusa, C. pisifera, Thuja standishii, Thujopsis dolabrata). Thuja plicata is highly revered among the tribes of the Northwest Coast of North America, who made their houses, canoes, baskets, boxes and even clothing from its bark, wood and roots, while Sequoia and Sequoiadendron have inspired a deep sense of reverence among people from western cultures ever since their discovery.
The Cupressaceae are also the most important conifer family in modern horticulture. Several species of Chamaecyparis, Cupressus and Juniperus are of major importance in horticultural trade, accounting for about 99.9% of all conifers sold for garden planting in Britain; many thousands of cultivars have been named.
Although the Pinaceae continue to account for the majority of dendrochronological work, a great deal has nonetheless been done with the Cupressaceae, particularly in the genera Fitzroya, Juniperus, Sequoiadendron, and Thuja.
Observations
See the genus and species accounts.
Remarks
Pollination usually occurs in late winter or spring but may occur anytime from late summer to early winter for some species of Juniperus. Seed maturation occurs in late summer or autumn. Many species of Callitris, Cupressus, Hesperocyparis, Sequoiadendron and Widdringtonia have serotinous cones that remain closed for many years, some opening only after exposure to fire (Watson and Eckenwalder 1993).
A majority of genera are monotypic and most others display disjunct or relictual distributions, even though individual species may be widely distributed. Only bird-dispersed Juniperus is species rich, with a wide, nearly continuous Northern Hemisphere distribution. Because of their uniformity, seedlings and juvenile specimens may not be determinable to genus (Watson and Eckenwalder 1993). Foliage of cultivars may deviate greatly from forms found in wild plants; one, cv. 'Sanderi', defied correct identification for 84 years until 1978 when chemical analysis discovered its generic and specific identity as a juvenile-foliage cultivar of Platycladus orientalis; it had previously been placed under five other genera (including one Shishinderia created for it alone!) by various authors (Gough and Welch 1978).
Although no members of the family attain dominance over immense geographic spans as do some species of the Pinaceae in the boreal forests, they can achieve considerable local and regional prominence. Examples include Sequoia sempervirens along the coast of northern California, several species of Juniperus in central Eurasia, the southwestern United States and Mexico, and baldcypress (Taxodium distichum) in deep swamps of the southeastern United States. Their ranges and regions of dominance were considerably greater during the early Tertiary (Watson and Eckenwalder 1993).
Citations
American Lumberman. 1911.11.11. "The Realization of a Great Commercial Dream." American Lumberman n.v. (November 11, 1911):43-142.
Brundrett, Mark. 2008. Mycorrhizal Associations: The Web Resource. mycorrhizas.info, accessed 2009.06.09.
Eckenwalder, J. E. 1976. Re-evaluation of Cupressaceae and Taxodiaceae: A proposed merger. Madroño 23:237-256.
Escapa, I. , R. Cúneo, and B. Axsmith. 2008. A new genus of the Cupressaceae (sensu lato) from the Jurassic of Patagonia: Implications for conifer megasporangiate cone homologies. Review of Palaeobotany and Palynology 151:110-122.
Gough, L. J. and H. J. Welch. 1978. Bot. J. Linn. Soc. 77:217-221.
Gray, S. F. 1822. Nat. Arr. Brit. Pl. 2:222, 225. Available: Biodiversity Heritage Library, accessed 2026.01.27.
IUCN. 2020. IUCN Red List version 2020-1: Table 4b: Red List Category summary for all plant classes and families. https://nc.iucnredlist.org/redlist/content/attachment_files/2020_1_RL_Stats_Table_4b.pdf, accessed 2022.09.23.
Li, Zheng, Anthony E. Baniaga, Emily B. Sessa, Moira Scascitelli, Sean W. Graham, Loren H. Rieseberg, and Michael S. Barker. 2015. Early genome duplications in conifers and other seed plants. Science Advances 1(10):e1501084.
Newman, E.I. and P. Reddell. 1987. The distribution of mycorrhizas among families of vascular plants. New Phytologist 106: 745-751.
Oersted. 1864. Vidensk. Meddel. Dansk Naturahist. Foren. Kjobenhavn, ser. 26: 32.
Rothwell, G. W., G. Mapes, R. A. Stockey, and J. Hilton. 2012. The seed cone Eathiestrobus gen. nov.: fossil evidence for a Jurassic origin of Pinaceae. American Journal of Botany 99(4):708-720.
Yang, Yong, David Kay Ferguson, Bing Liu, Kang-Shan Mao, Lian-Ming Gao, Shou-Zhou Zhang, Tao Wan, Keith Rushforth, and Zhi-Xiang Zhang. 2022. Recent advances on phylogenomics of gymnosperms and a new classification. Plant Diversity 44(4):340-350.
See also
Burns and Honkala (1990).
Canadian Forestry Service. 1983. Reproduction of conifers. Forest. Techn. Pub. Canad. Forest. Serv. 31.
Farjon (2005).
Michael P. Frankis contributed greatly to developing this page, 1999.02.
Farjon (2005).












